Atomic Make-up
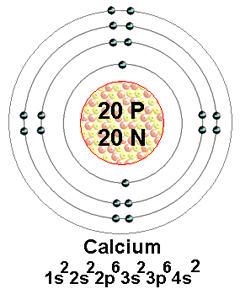
Protons = 20
Neutrons= 20
Electrons= 20
Neutrons= 20
Electrons= 20
The Electron configuration notation for Calcium is 1s2 2s2 2p6 3s2 3p6 4s2= [Ar]
What is the ore?

- The ore of Calcium is Calcium Oxide (also known as quiklime).
- Calcium Oxide is a mixture of lime and mercuric oxide
- Calcium oxide is usually made by the thermal decomposition of materials such as limestone, process called calcination or lime-burning ( Calcium Oxide, 24.04.12, en.wikipedie.org).
- Calcium Oxide is a mixture of lime and mercuric oxide
- Calcium oxide is usually made by the thermal decomposition of materials such as limestone, process called calcination or lime-burning ( Calcium Oxide, 24.04.12, en.wikipedie.org).
Where is it located?
- Calcium is found in chalk, limestone, gypsum and anhydrite.
- Most of Calcium oxide is mined in America.
- Also is located in countries such as North America, South America, Eastern Europe, South East Asia, Africa, Oceania, Mid East, Eastern Asia and Western Europe.
- Most of Calcium oxide is mined in America.
- Also is located in countries such as North America, South America, Eastern Europe, South East Asia, Africa, Oceania, Mid East, Eastern Asia and Western Europe.
How Calcium Oxide is mined...
Calcium metal was not available in a large scale until the beginning of the 20th century. Calcium oxide (CaO) is a white solid rock and is made by heating calcium carbonate (CaCO3) at high temperatures.
This is the chemical equation: CaCo3 -> Co2 + Cao.
Industrially it is used to treat acidic soil and to make porcelain and glass, bleaching powder, caustic soda, mortar and cement. Calcium oxide reacts with water to form calcium hydroxide (Ca(OH)2). There is not much demand for pure calcium. Most calcium is used in the form of limestone, gypsum, or other minerals that can be mined directly from the earth.
This is the chemical equation: CaCo3 -> Co2 + Cao.
Industrially it is used to treat acidic soil and to make porcelain and glass, bleaching powder, caustic soda, mortar and cement. Calcium oxide reacts with water to form calcium hydroxide (Ca(OH)2). There is not much demand for pure calcium. Most calcium is used in the form of limestone, gypsum, or other minerals that can be mined directly from the earth.
Are there any issues that can be dangerous to our culture?

Calcium Oxide is part of the alkaline earth metals which are a reducing agent. All alkaline earth metals react with such elements as oxygen and chorine which becomes more vigorous as their atomic number increases. Many of the metals produce a characteristic colored flame when they burn. Calcium turns into the colour brick-red.
This can be dangerous when mining these metals as they could react with other elements. In the year 2007, Portland cement was in seriously short supply in the Nation. The cement shortages also caused construction delays that resulted in increased costs for roads, bridges, and buildings.
This can be dangerous when mining these metals as they could react with other elements. In the year 2007, Portland cement was in seriously short supply in the Nation. The cement shortages also caused construction delays that resulted in increased costs for roads, bridges, and buildings.
How Calcium is used commercially, militarily and scientifically:
Calcium oxide is used as an alkali for treating acidic soils and in manufacturing steel, paper, and glass, and it is the main component of lime. Commercial heat sources for self-heating food packaging use an exothermic reaction between quicklime, or calcium oxide, and water, which generates 60 calories of heat per gram (heating-food-packaging, wikipedia.com, 29/04/12). Quicklime is inexpensive and readily available, which is recognised by the FDA as being safe to use. The by-product of the reaction is calcium hydroxide. Calcium oxide has been used for hundreds of years in construction, sculptures, and roads. It is so useful, that it is today produced industrially on a vast scale; over 20 million metric tons were produced in the U.S. in 2000. They include chalk, gypsum, limestone, marble, and plaster of paris.
